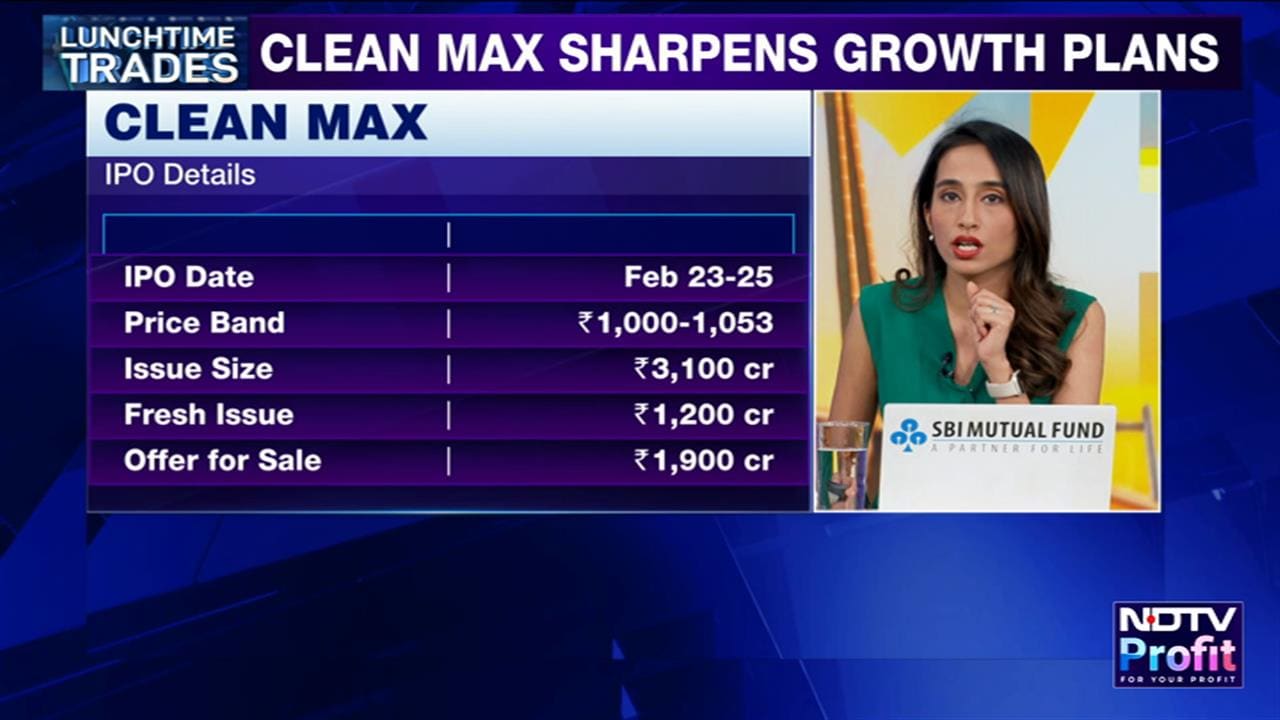Bajaj Healthcare on Thursday said it has received approval from the Drug Controller General of India to manufacture a drug for the treatment of hallucinations and delusions associated with Parkinson's disease.
The company has received approval to produce both the API and drug formulation of Pimavanserin, a 34 mg capsule.
It is an atypical antipsychotic used for the treatment of hallucinations and delusions associated with Parkinson's disease psychosis.
Pimavanserin, marketed globally as Nuplazid, has become a preferred treatment in the US antipsychotic segment.
In a recent update, Acadia Pharmaceuticals announced that Nuplazid, along with its other brand Daybue, is projected to generate more than $1 billion in net sales in 2025.
"The growing success of Nuplazid in the US underscores the global demand for this innovative therapy, and we are confident that its introduction in India will be a transformative advancement in the antipsychotic segment," Bajaj Healthcare Managing Director Anil Jain said.
The company said it has also extended offer for Pimavanserin to several leading Indian pharmaceutical companies, ensuring the product's availability in the domestic market.
Essential Business Intelligence, Continuous LIVE TV, Sharp Market Insights, Practical Personal Finance Advice and Latest Stories — On NDTV Profit.