
The U.S. drug shortage was at its worst since 2014 in the quarter ended March. While that should suggest better demand and pricing for Indian drugmakers battling competition in the American market, pharma companies and analysts see limited upside.
Sun Pharmaceuticals Industries Ltd., India's largest drugmaker, said it has "not seen price stabilisation" in the period. This, the company's management said in its fourth-quarter earnings call, is "not just from Sun's perspective, but also from an industry perspective".
According to Manoj Garg, director of investments at WhiteOak Capital Management, the intensity of price erosion may come down compared to the recent past. But companies will not be able to take price hikes, except for marginal increases in a few products where there are shortages in the market, he said.
That suggests no relief for Indian makers of copycat drugs in one of their biggest markets. Domestic companies have been grappling with severe price erosion for the past couple of years, leading to compressed U.S. margins and profitability. Increased scrutiny on manufacturing standards has only made it worse.
U.S. Drug Dearth
According to the American Society of Health-System Pharmacists, drug shortages in the U.S. "are not resolving and new shortages are increasing. Ongoing and active shortages are the highest since 2014".
A five-year trend of quarterly data released by ASHP indicates how drug shortages peaked in the last quarter. Local anesthetics, and basic hospital drugs, albuterol solution, common oral and ophthalmic products, ADHD treatments and chemotherapy drugs without alternatives are in short supply, ASHP noted.
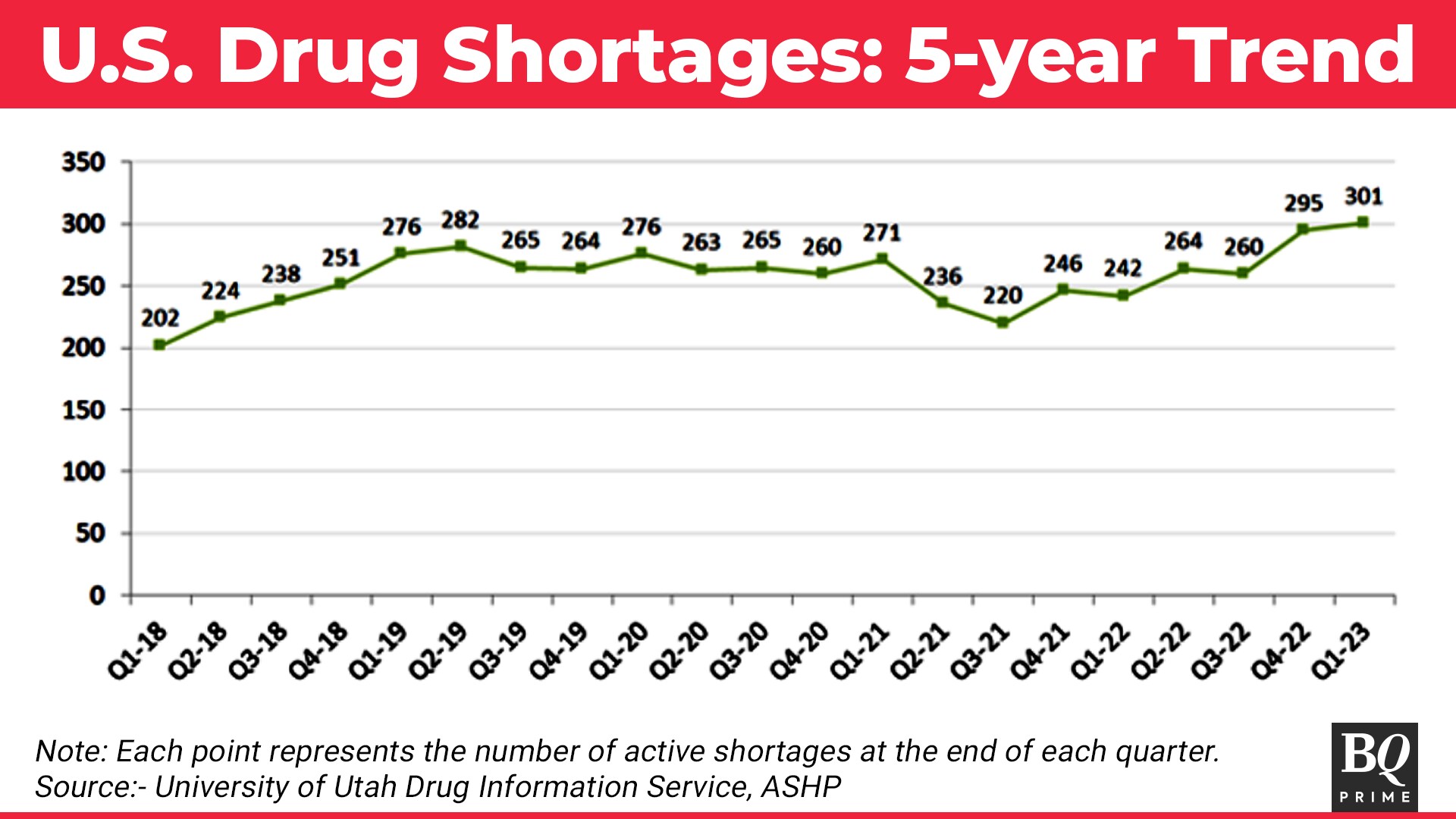
Market Dynamics
The shortage of medicines in the U.S. has been a constant problem and has aggravated since 2020, Salil Kallianpur, pharma marketing and strategy consultant, told BQ Prime.
"This is clearly due to offshoring manufacturing and relying on a global manufacturing and supply chain for even basic medicines. Ideally, India, which supplies 80% of intermediates to the U.S. (along with China), should have benefitted from this. Even more because low priced generic medicines are the most affected in short supply."
But Food and Drug Administration audits highlighting lapses in quality standards in companies that have very large supply arrangements in the U.S. have been causing temporary delay in supplies, aggravating drug shortages.
Also, price consolidation in the U.S. is still ongoing, Kallianpur said.
The generics market in the U.S. has been consolidated to few large distributors. Hence, they have higher bargaining power compared to drugmakers, Garg explained. This has led to price competition among manufacturers and suppliers.
Structurally, the U.S. is a highly commoditised market and more competition is only leading to further pricing pressure, Garg said.
In the past, only one company that was first-to-file enjoyed exclusivity over the generic post patent-expiry. But now, the U.S. FDA allows all companies that have filed on the first day to be FTFs, leading to competition and price margin erosion from day one, said Aditya Khemka, fund manager at InCred PMS Healthcare.
Marginal Upside
In the past, drug shortages were a meaningful indicator for price hikes, said Khemka. They allowed pharma companies to increase prices of scarce drugs by 10-100% and even as high as 15 to 20 to 100 times.
However, in 2015, the Federal Trade Commission in the U.S. penalised companies for colluding and hiking prices on scarce drugs. Companies under the scanner paid penalties in 2019, said Khemka.
This time, while there may be no further price erosion on drugs which are in short supply, he said, companies may choose to take only marginal price hikes because of past penalties.
Also, Khemka said, out of 16,000 stock-keeping units being sold in the U.S., the shortage is being experienced for 240 to 250 SKUs and therefore, the overall impact on prices may not be material.
It is early to say if prices would go back to earlier levels, according to Kallianpur. "At best, marginal improvement can be expected in this fiscal."
The pricing pressure which was in mid-to-high teens in the past two to three years has come down to mid-to-high single digit, said WhiteOak Capital's Garg. For highly commoditised portfolios, while the prices have hit rock bottom and may not see further erosion, companies may not be able to take further hike except on a few products, he said.
Essential Business Intelligence, Continuous LIVE TV, Sharp Market Insights, Practical Personal Finance Advice and Latest Stories — On NDTV Profit.






















